Background: For patients with relapsed/refractory large B-cell lymphomas (rrLBCL), CD19-directed chimeric antigen receptor T cells (CAR19) improve survival compared to autologous hematopoietic cell transplantation (HCT). However, major toxicities of CAR19 therapy include prolonged cytopenias and associated infections. To better understand the impact of CAR19 on such toxicities, we studied a cohort of LBCL patients achieving durable remissions to assess immune recovery after CAR19 treatment. We (1) compared immune reconstitution in HCT vs CAR19 treated patients, (2) performed molecular analysis of post-CAR19 cytopenic marrow specimens, and (3) profiled emergent mutations in cell free DNA (cfDNA) before and after CAR19.
Methods & Results: We first propensity matched rrLBCL patients treated at Stanford with either CAR19 (100% axi-cel) or HCT who achieved durable remissions. Patients were matched based on age, sex, prior systemic lines of therapy, and time from infusion. From 471 eligible cases, propensity score matching was successful in 138 (n=69 per arm). When compared to HCT-treated counterparts, CAR19-treated patients had reduced recovery of absolute lymphocyte counts (ALC) and absolute neutrophil counts (ANC; longitudinal regression day 28-800 ANOVA p<0.005), but similar recovery of hemoglobin and platelets. CAR19-treated patients experienced significantly more Grade ≥3 infections in competing risk analyses (HR 2.61, CI 1.26-5.42), and higher non-relapse mortality (log-rank p=0.027).
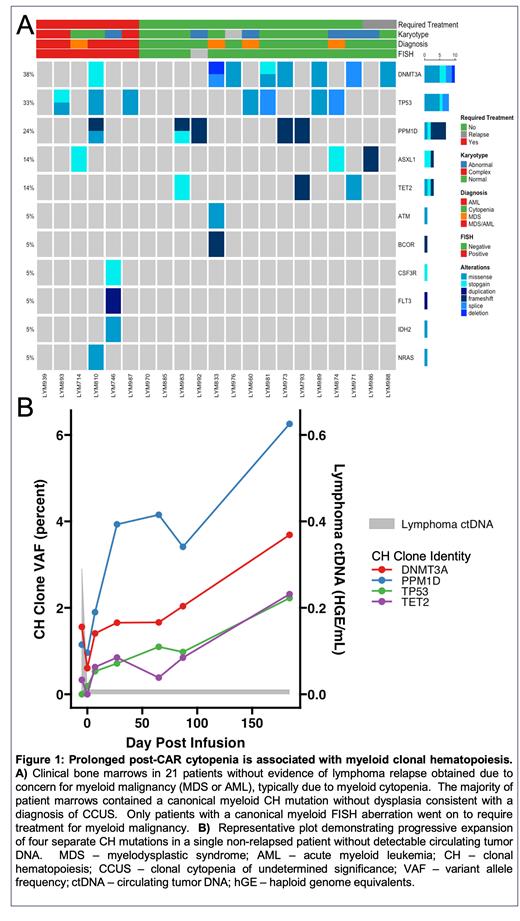
Next, to characterize mechanisms underlying cytopenias after CAR19, we profiled marrow aspirates from cases requiring biopsy due to prolonged cytopenia or clinical concern for myeloid neoplasms. No patient had evidence of lymphoma relapse at the time of biopsy, and all were >60d from CAR19 infusion (median 377d, range 86-1561d). Among 21 patients profiled by DNA sequencing, 18 (86%) had evidence of ≥1 somatic mutation in genes canonically associated with clonal hematopoiesis (CH) in the marrow ( Fig. 1A), with the most recurrently mutated genes being DNMT3A (38%), TP53 (33%), PPM1D (24%), ASXL1 (14%), and TET2 (14%). Among 6 patients diagnosed with and requiring therapy for a myeloid malignancy, all (100%) had a canonical myeloid karyotypic aberration detected by FISH, as compared with 0 in the 14 other evaluable cases ( Fig. 1A). CAR19 cells were proportionately enriched in the marrow relative to paired peripheral blood by flow cytometry (n = 12, median 2.5-fold enrichment, Wilcoxon p<0.001). Using 10x scRNA-Seq, viable CAR19 was detectable in 7 of 8 marrows (88%) and were predominantly CD4+.
We hypothesized that CH clones detectable after CAR19 would also be present in pre-CAR19 blood samples and detectable in cfDNA. To test this, we profiled 786 longitudinal samples from 141 patients using CAPP-seq and serially monitored CH clones in cfDNA. Among 141 eligible cases, 40 (28%) were evaluable for CH, in having both pre- and post-CAR19 infusion specimens, while in durable remission (median follow up 967d). Of 51 CH clones detected in post-infusion cfDNA, 46 (90.1%) were also detectable prior to CAR19, typically expanding post-infusion (median = 1.8-fold, Fig. 1B).
We next tested whether expanding CH clones showed evidence for selection after CAR19. Strikingly, among 48 recurrently mutated genes tested, DNMT3A and PPM1D showed significant evidence for clonal selection and expansion after CAR19 (Q-value < 0.05). When compared to patients without emerging CH clones, patients with ≥5-fold clonal expansions of canonical CH genes (23% of cases) had reduced ANC, but not ALC, recovery (median ANC=1.43 vs 2.43 at 6-mo, Wilcoxon p<0.001). This association remained significant in multivariate longitudinal regression that included age, sex, prior lines of therapy, and pre-infusion ANC (ANOVA p = 0.007).
Conclusion: CAR19 treated patients experience reduced immune reconstitution and higher infection rates than HCT treated counterparts. Bone marrow findings reveal prolonged CAR19 cytopenia to commonly be associated with clonal cytopenia of undetermined significance (CCUS). Despite durable lymphoma remissions, canonical myeloid clonal expansions are common post-CAR19, and associated with myelosuppression. Non-invasive genotyping could aid in early identification of patients at risk for prolonged cytopenias after CAR19 and inform interventions to decrease associated complications.
Disclosures
Hamilton:Kite Pharma: Other: Advisory Board. Sworder:Foresight Diagnostics: Consultancy. Alig:Takeda: Honoraria. Shahrokh Esfahani:Foresight Diagnostics: Consultancy. Muffly:amgen: Consultancy; adaptive: Membership on an entity's Board of Directors or advisory committees, Research Funding; orca bio: Research Funding; jasper: Research Funding; kite: Consultancy, Honoraria, Research Funding; pfizer: Consultancy; bms: Research Funding; autolus: Consultancy; astellas: Consultancy, Research Funding. Negrin:Garuda Therapeutics: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Amgen: Membership on an entity's Board of Directors or advisory committees; Co-Immune: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Appia Bio: Membership on an entity's Board of Directors or advisory committees; Regimmune, Inc.: Consultancy; UpToDate: Patents & Royalties; Orca Bio: Research Funding; Cellenkos: Consultancy; Biorasi: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees. Lowsky:Orca Bio: Research Funding. Meyer:Orca Bio: Research Funding. Rezvani:Pharmacyclics.: Research Funding. Shizuru:Jasper Therapeutics: Consultancy, Current equity holder in publicly-traded company. Sidana:Magenta Therapeutics, BMS, Janssen, Sanofi, Oncopeptides, Takeda, Pfizer: Consultancy; Magenta Therapeutics, BMS, Allogene, Janssen, Novartis: Research Funding. Smith:A28: Membership on an entity's Board of Directors or advisory committees; BMS: Consultancy. Dahiya:Adaptive Biotechnologies: Consultancy; Incyte: Consultancy; Bristol Myers Squibb: Consultancy; Kite, a Gilead Company: Consultancy, Research Funding. Diehn:Stanford University: Patents & Royalties: ctDNA detection, tumor treatment resistance Mechanisms; Boehringer Ingelheim: Consultancy; Genentech: Consultancy, Research Funding; Novartis: Consultancy; BioNTech: Consultancy; Gritstone Bio: Consultancy; Illumina: Consultancy, Research Funding; AstraZeneca: Consultancy, Research Funding; Roche: Consultancy; CiberMed: Current holder of stock options in a privately-held company; Varian Medical Systems: Research Funding; Genentech: Consultancy, Research Funding; Varian Medical Systems: Research Funding; Stanford University: Patents & Royalties: ctDNA detection, tumor treatment resistance Mechanisms; Foresight Diagnostics: Current Employment, Current holder of stock options in a privately-held company; Boehringer Ingelheim: Consultancy. Frank:Roche/Genentech: Current holder of stock options in a privately-held company; Allogene: Consultancy; Kite, a Gilead Company: Research Funding; Adaptive Biotechnology: Consultancy; EcoR1: Consultancy; BRVLH: Consultancy; Cargo Therapeutics: Consultancy, Other: Travel Support; Gilead Sciences: Consultancy, Other: Travel Support. Mackall:Mammoth: Consultancy, Current equity holder in private company; Lyell Immunopharma: Current equity holder in private company, Research Funding; Immatics: Consultancy; CARGO: Consultancy, Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; Adaptimmune: Consultancy; Link Cell Therapies: Consultancy, Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees. Kurtz:Foresight Diagnostics: Consultancy, Current equity holder in private company, Current holder of stock options in a privately-held company, Patents & Royalties: Patents Pertaining to circulating tumor DNA licensed to Foresight Diagnostics. Miklos:NA: Patents & Royalties: cGVHD patent holder for Ibrutinib as cGVHD therapy but no compensation; A2 Biotherapeutics: Consultancy, Current holder of stock options in a privately-held company, Honoraria; Amgen: Consultancy, Honoraria; 2Seventy Bio: Research Funding; Adicet: Research Funding; Bristol-Myers Squibb: Consultancy; Allogene: Research Funding; Miltenyi: Consultancy, Research Funding; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Genentech: Consultancy, Honoraria; Fate Therapeutics: Research Funding; Adaptive Biotechnologies: Consultancy; Kite, a Gilead Company: Consultancy, Research Funding; Gilead Sciences: Consultancy, Honoraria; Incyte: Consultancy, Honoraria; Juno Therapeutics: Consultancy, Honoraria, Patents & Royalties: rights to royalties from Fred Hutch for patents licensed to Juno, Research Funding; Legend Biotech: Consultancy, Honoraria; MorphoSys: Consultancy, Honoraria; Mustang Bio: Consultancy, Honoraria; Navan Technologies: Consultancy, Current holder of stock options in a privately-held company, Honoraria; Novartis: Consultancy, Honoraria; Pharmacyclics: Consultancy, Honoraria; Umoja: Consultancy, Honoraria; Bioline Rx: Membership on an entity's Board of Directors or advisory committees; Janssen: Consultancy, Honoraria, Other: Travel support. Alizadeh:Roche: Consultancy, Honoraria, Other: Travel, accommodations and expenses; Celgene: Consultancy, Research Funding; Janssen Oncology: Honoraria; Foresight Diagnostics: Consultancy, Current holder of stock options in a privately-held company; Syncopation Life Sciences: Current holder of stock options in a privately-held company; Forty Seven: Current holder of stock options in a privately-held company; Lymphoma Research Foundation: Consultancy; Stanford University: Patents & Royalties: ctDNA detection; CiberMed: Consultancy, Current holder of stock options in a privately-held company; CAPP Medical: Current holder of stock options in a privately-held company; Gilead Sciences: Consultancy, Other: Travel, accommodations and expenses.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal